Anomalous diffusion of confined long-chain molecules: the thermal resistance effect
17 May 2021
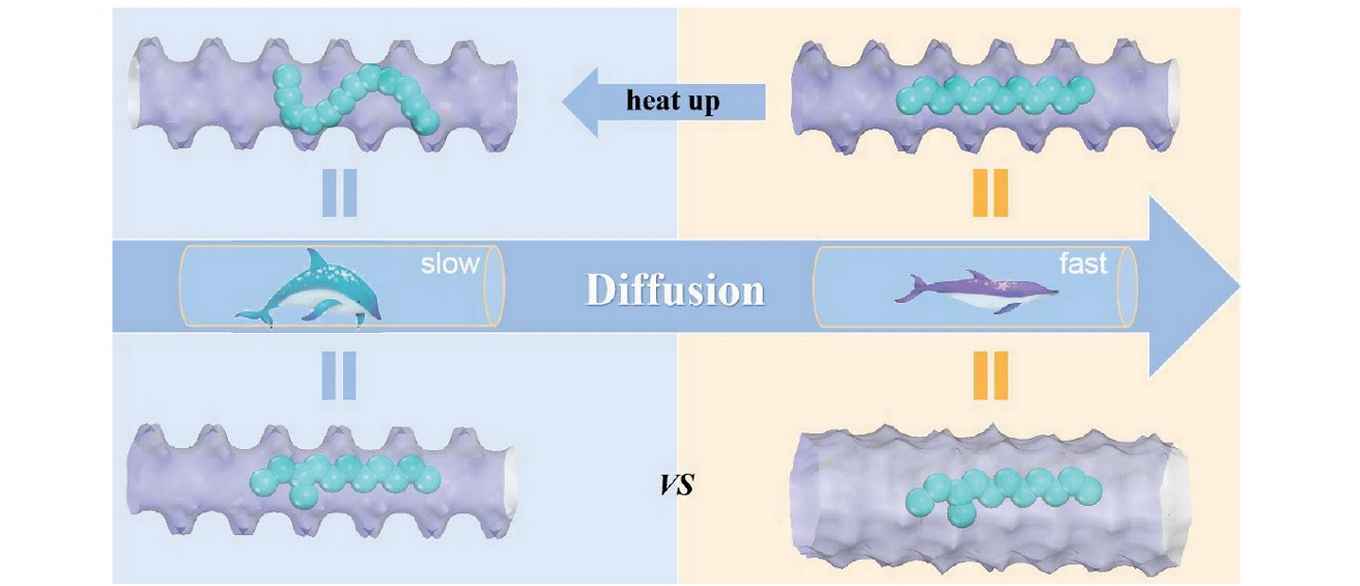
The research was performed by researchers at laboratories and institutes of the Chinese Academy of Sciences in Wuhan, Bejing and Dalian, together with the Shanghai Research Institute of Petrochemical Technology. Prof. Rajamani Krishna of the University of Amsterdam's Van 't Hoff Institute for Molecular Sciences contributed with ab initio molecular dynamics (MD) simulations that clearly established the connection between the molecular structure and anomalous diffusion.
Abstract
Diffusion is generally faster at higher temperatures. Here, a novel counterintuitive behaviour is observed that the movement of long-chain molecules slows as the temperature increases under confinement. The report confirms that this anomalous diffusion is caused by the “thermal resistance effect” where the diffusion resistance of linear-chain molecules is equivalent to that with branched-chain configurations at high temperature. It then restrains the molecular transportation in the nanoscale channels as further confirming by zero length column experiments. This work enriches the anomalous diffusion family and provides fundamental insights for the novel mechanism inside confined systems.
Paper
Jiamin Yuan, Zhiqiang Liu, Yimo Wu, Jingfeng Han, Xiaomin Tang, Chengbin Li, Wei Chen, Xianfeng Yi, Jian Zhou, Rajamani Krishna, German Sastre, Anmin Zheng: Thermal resistance effect on anomalous diffusion of molecules under confinement. PNAS May 17, 2021 DOI: 10.1073/pnas.2102097118