New metal-organic framework for highly efficient separation of ethane from ethylene
Industrial application could make manufacturing of plastics more energy-efficient
29 October 2018
Making plastic takes lots of energy. Polyethylene, the most common type of plastic, is built from ethylene, one of the many hydrocarbon molecules found in crude oil refining. The ethylene must be highly purified for the manufacturing process to work, but the current industrial technology for separating ethylene from all the other hydrocarbons is a high-energy process that cools down the crude to more than 100 degrees below zero Celsius.
Ethylene and ethane constitute the bulk of the hydrocarbons in the mixture, and separating these two is by far the most energy-intensive step. Finding an alternative method of separation would reduce the energy needed to make the 170 million tons of ethylene manufactured worldwide each year. Microporous materials such as metal-organic frameworks (MOFs) can hold the key to energy efficient separation as they can be tuned to preferentially bind one component over another.
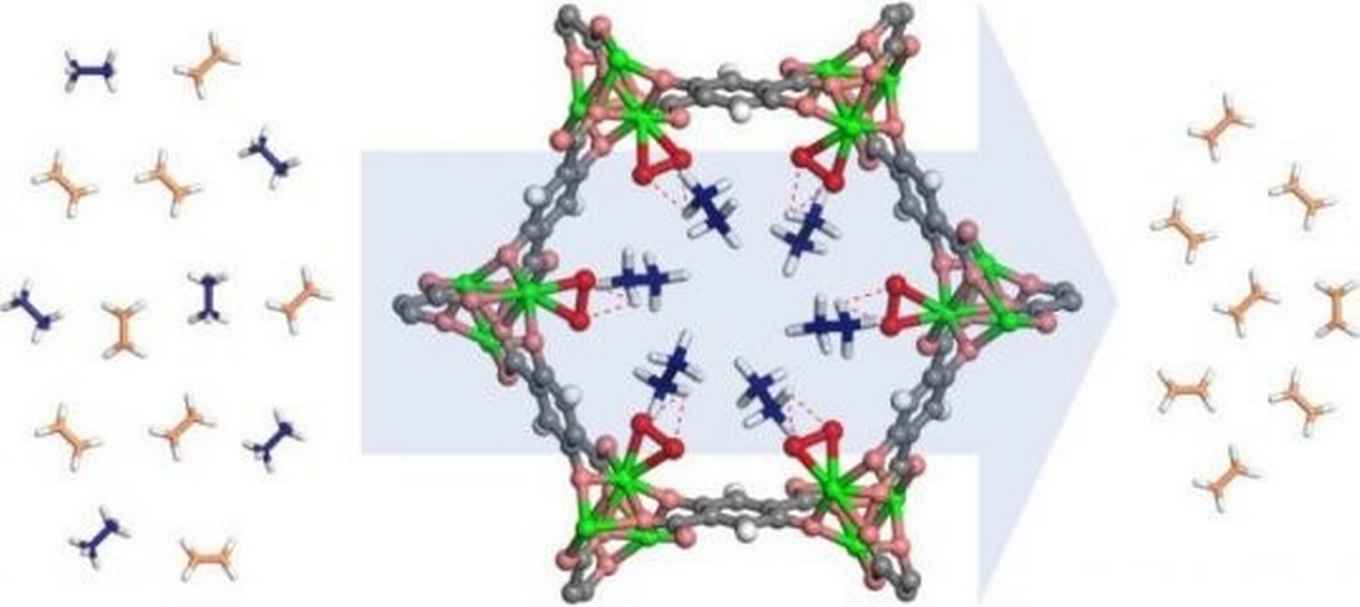
Purify ethylene out of a mixture with ethane
In the current issue of Science the Chinese-American research team now reveals that a modification to a well-studied MOF enables it to separate purified ethylene out of a mixture with ethane. The MOF was created at The University of Texas at San Antonio (USA) and China's Taiyuan University of Technology and studied at the NIST Center for Neutron Research (NCNR) in Gaithersburg, USA. The authors show its ability to produce polymer-grade ethylene (99.99%) from a single cycle separation cycle at ambient conditions.
Prof. Rajamani Krishna of the University of Amsterdam's Van 't Hoff Institute for Molecular Sciences contributed to this investigation by performing detailed comparisons of the separation performance with competing MOFs. These comparisons were performed using transient breakthrough simulations using the methodology described by Krishna in 'Screening Metal-Organic Frameworks for Mixture Separations in Fixed-Bed Adsorbers using a Combined Selectivity/Capacity Metric', published in 2017 in RSC Advances.
Publication details
Libo Li, Rui-Biao Lin, Rajamani Krishna, Hao Li, Shengchang Xiang, Hui Wu, Jinping Li, Wei Zhou, Banglin Chen: Ethane/ethylene separation in a metal-organic framework with iron-peroxo sites. Science 26 Oct 2018 Vol. 362, Issue 6413, pp. 443-446. DOI: 10.1126/science.aat0586