Temperature- and light-responsive switch for acetylene/ethylene separation
HIMS researcher Rajamani Krishna contributes to international research project
25 April 2017
Metal Organic Frameworks (MOF) are an emerging class of inexpensive materials with great potential for energy efficient separation of gaseous mixtures. MOFs consist of metal ions combined with organic linker molecules to form highly structured porous molecular architectures. The pores can be straightforwardly and rationally tuned to enforce size selective sieving effects. Furthermore the pore surfaces can be readily functionalized to induce preferential interactions with specific molecules.
Professor Krishna of the Computational Chemistry research group at HIMS has performed numerous simulations of the performance of MOFs in a broad range of potential applications. This follows from his continuous focus on improving technologies related to reaction and separation. He is an internationally recognized expert in the investigation of physico-chemical phenomena at the molecular and microscopic levels.
Smart molecular switch
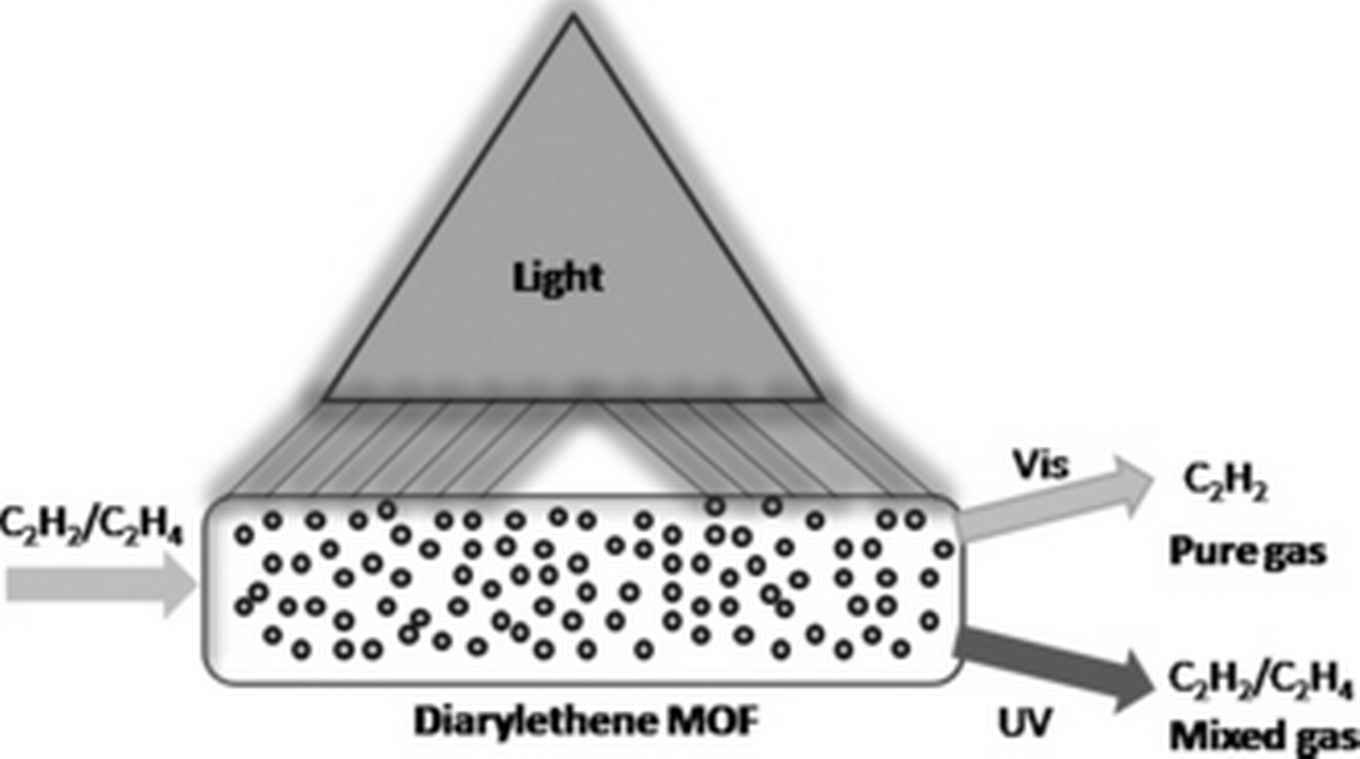
Diarylethene has been extensively explored in photochemistry and material science, since it undergoes a fast, reversible photoinduced ring-open to ring-closed transformation and has high photostability for each isomer. Since such isomerization usually results in significant difference in optical and electronic properties its application as molecular switching material was recognized before. Inspired by this, the use of photochromic diarylethene to switch gas separation was subject to the described research project.
Krishna and colleagues discovered that diarylethene incorporated in a metal–organic framework (MOF) give temperature- and light-responsive switch properties to the separation material. For the industrial very relevant process of separation of acetylene/ethylene (C2H2/C2H4) separation this material shows ultrahigh C2H2/C2H4 selectivity of 47.1 at 195 K. This is over 20 times larger than the value at 293 K, or over 15 times larger than the value of at 195 K under 365 nm UV light irradiation (all at 100 kPa). The origin of this unique control in C2H2/C2H4 selectivity, as unveiled by density functional calculations, is due to a guest discriminatory gate-opening effect from the diarylethene unit.
Article
C.B. Fan, L.L. Gong, L. Huang, F. Luo, R. Krishna, X.F. Yi, A.M. Zheng, L. Zhang, S.Z. Pu, X.F. Feng, M.B. Luo, G.C. Guo, Significant Enhancement of C2H2/C2H4 Separation by a Photochromic Diarylethene Unit: A Temperature- and Light-Responsive Separation Switch, Angew. Chem. Int. Ed, 24 April 2017.