Photocatalytic azolation of arenes using heterogeneous carbon nitride
ChemSusChem pick of the week
24 November 2021
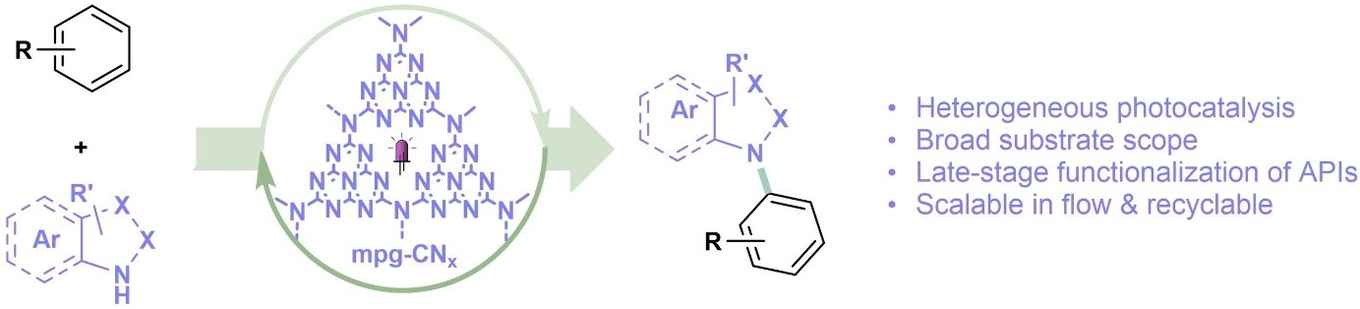
This strategy exhibits a broad substrate scope and is amenable to the late-stage functionalization of several pharmaceuticals. The catalyst can be easily recovered and reused leading to greener and more sustainable routes for the synthesis of compounds that are of relevance to pharmaceutical and agrochemical research.
Abstract
The functionalization of aryl C(sp2)−H bonds is a useful strategy for the late-stage modification of biologically active molecules, especially for the regioselective introduction of azole heterocycles to prepare medicinally-relevant compounds. Herein, we describe a practical photocatalytic transformation using a mesoporous carbon nitride (mpg-CNx) photocatalyst, which enables the efficient azolation of various arenes through direct oxidation. The method exhibits a broad substrate scope and is amenable to the late-stage functionalization of several pharmaceuticals. Due to the heterogeneous nature and high photocatalytic stability of mpg-CNx, the catalyst can be easily recovered and reused leading to greener and more sustainable routes, using either batch or flow processing, to prepare these important compounds of interest in pharmaceutical and agrochemical research.
Publication details
Zhenghui Wen, Ting Wan, Arjun Vijeta, Carla Casadevall, Laura Buglioni, Erwin Reisner, Timothy Noël: Photocatalytic c−h azolation of arenes using heterogeneous carbon nitride in batch and flow. ChemSusChem, First published: 16 September 2021 DOI: 10.1002/cssc.202101767