Performance of an electrocatalyst for converting CO2 captured from air
14 January 2026
Combining CO2 capture and utilization requires the development of highly selective catalysts to convert CO2 into valuable chemicals. While many studies focus on converting purified and concentrated CO2, the CO2 that has been obtained through direct air capture (DAC) can contain up to 10% oxygen. In the best case, the oxygen reduces the CO2 conversion efficiency. In the worst case, reactive oxygen species (ROS) are generated that lead to catalyst degradation.
In a joint project with Skytree, one of the leading companies in the Netherlands focusing on direct air capture of CO2, the HIMS researchers aim to develop robust catalysts that are instrumental to valorising air-captured CO2. In this, the research now published in JACS represents the first step. The project was co-funded by the Duch Ministry of Economic Affairs and Climate through the Top Consortia for Knowledge and Innovation (TKI) scheme.
Mechanism dependent oxygen tolerance
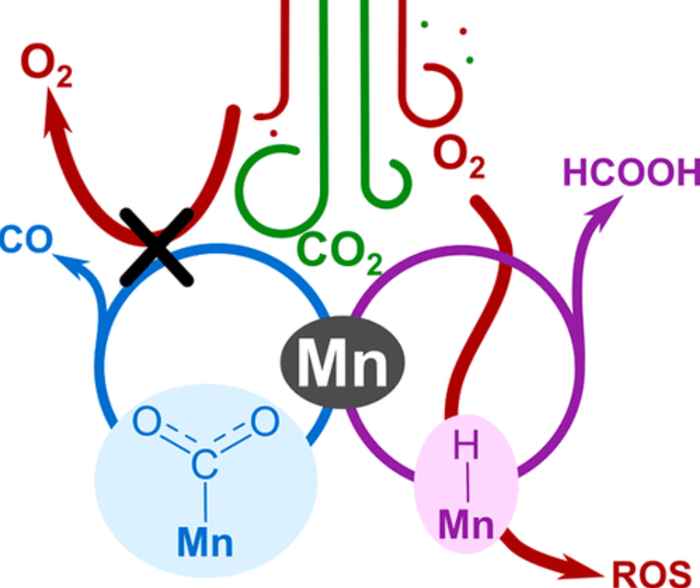
The research mapped the intrinsic reactivity of a homogeneous manganese-based catalyst under DAC-mimicking conditions. Depending on reaction conditions, this catalyst can selectively form either CO or formic acid (HCOOH). The researchers found the oxygen tolerance of the catalyst to be dependent on the associated reaction mechanism. For conversion towards CO, the selective mechanism is CO2 binding and since this is faster than O2 binding, the mechanism is intrinsically oxygen-tolerant. On the other hand, the conversion towards formic acid goes via a manganese-hydride intermediate. The research shows that for this mechanism, oxygen can severely hamper the CO2 conversion performance.
Based on these insights, the team headed by Dr Pullen will now develop novel catalysts that can convert DAC CO2 without suffering from lower stability and conversion efficiency.
Paper details
Catriona C. James, Maartje C. van Rijn, and Sonja Pullen: Mechanism-Dependent Oxygen Tolerance of Mn(bpy)CO3Br in Electrochemical CO2 Reduction. Journal of the American Chemical Society. Article ASAP, January 8, 2026. DOI: 10.1021/jacs.5c17737