Novel iron-based catalyst for carbene radical chemistry
20 February 2026
Carbene-radical transfer reactions are widely used to facilitate a range of industrially relevant chemical conversions. Until now, this required the use of catalysts that are mainly based on precious metals or transition metals. In search of a readily available, cost-effective, and environmentally benign alternative, a Chinese research team, led by Prof. Xinfang Xu at Zhejiang Sci-Tech University in Hangzhou, set out to explore the use of an iron-based catalyst.
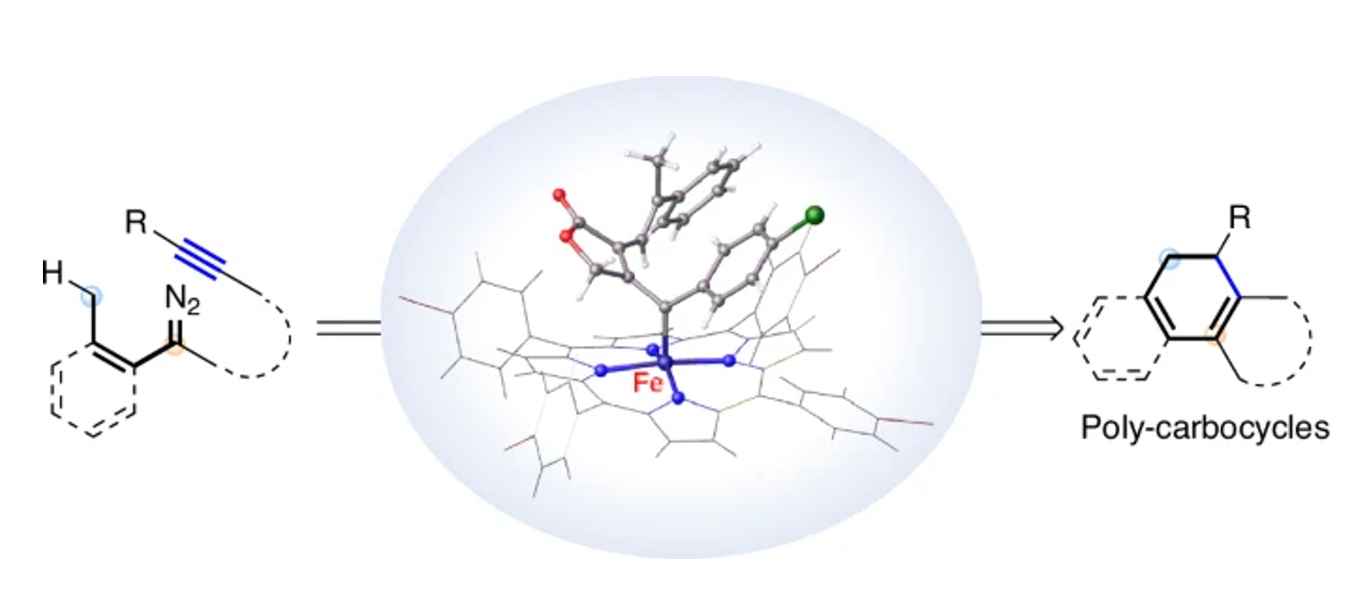
The Nature Catalysis paper demonstrates, for the first time, that an iron-based catalyst can indeed enable reactions involving carbene radicals. It presents examples of the synthesis of exo-cyclically substituted cyclohexadienes, which are important for the efficient synthesis of medicines and other bioactive compounds such as fragrances and flavourings. Furthermore, such compounds can be easily oxidised to yield their aromatic analogues. These triarylenes, featuring unique substitution patterns, are also very difficult to synthesise along other routes.
HIMS researchers provide mechanistic insight
At the Van ‘t Hoff Institute for Molecular Sciences, the research group of Prof. Bas de Bruin has long been at the forefront of carbene-radical chemistry. Inspired by the results obtained in Amsterdam over the past years, the Chinese researchers now explored new related reactions. They also focused on iron catalysts instead of the cobalt-based catalysts that have been the main focus of the Amsterdam research.
The group of Prof. Xinfang Xu approached the group of De Bruin to collaborate on this topic and explore the mechanism of the new iron-catalysed reactions. De Bruin teamed up with his HIMS colleague, Andreas Ehlers, to conduct a computational analysis of the reaction pathways. Corroborated by the Chinese experimental and analytic results, the Nature Catalysis paper now presents solid evidence of the mechanistic aspects of the iron-catalysed carbene transfer reactions. With these detailed mechanistic insights, novel iron-promoted carbene transfer reactions can be envisioned in the foreseeable future.
Abstract of the paper
Transition metal-catalyzed carbene transfer reactions are some of the most widely used methods that facilitate a range of otherwise inaccessible chemistry. While these species are generally promoted by precious metal catalysts, using inexpensive and less toxic iron complexes is under development. Moreover, surprisingly little is known about the mechanistic aspects, in particular the structures of these intermediates. Herein, we report an iron-catalyzed cascade reaction of alkyne-tethered diazo compounds, offering an efficient approach for the synthesis of carbocyclic molecules with structural diversity and flexibility under mild conditions. Control experiments and density functional theory calculations unambiguously reveal two distinct reaction pathways catalyzed by either Fe(II) or Fe(III) porphyrin complexes, which involve carbene and carbene radical intermediates, respectively. The structure of the key vinyl iron carbene intermediate has been determined by X-ray diffraction. Moreover, the utilization of a heterogeneous iron catalyst, FeP-CMP, which retains its catalytic efficacy even after being recycled ten times, facilitates a synthesis on 10 mmol scale with up to TON = 1820 and TOF = 303 h-1. The synthetic utility has been demonstrated by synthesis of poly-substituted arenes via a streamlined one-pot process.
Paper details
Xinke Zhang, Minghan Yao, Kewei Chen, Yuecheng Weng, Wenhua Zhang, Andreas W. Ehlers, Bas de Bruin & Xinfang Xu: Iron-catalyzed carbene and carbene radical cascade reactions for the synthesis of carbocyclic molecules. Nature Catalysis, 2026, DOI: 10.1038/s41929-026-01496-w