The physical mechanisms of nanoparticle–membrane interactions
Towards a rational design of nanoparticles for applications in medicine and adaptive materials
16 March 2026
Instead of relying on trial and error, nanoparticle developers can use the presented framework to understand how the combination of shape, flexibility and interaction of the particles can yield desired features.
This is, for instance, relevant in the field of nanocarriers for drug delivery and related biomedical applications. Understanding the effect of nanocarrier flexibility, topology and adhesion on membrane wrapping may help guide the development of carriers with improved targeting efficiency and enhanced cellular uptake.
Since the framework is modular and tuneable, it can be extended to study particles whose mechanical or surface properties adapt to local conditions, opening opportunities not only in nanomedicine but also in the design of bioinspired and responsive soft materials.
Abstract, as published with the paper
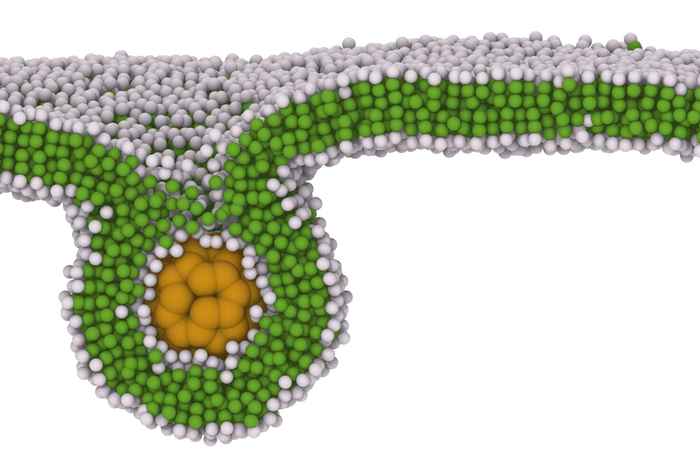
Nanoparticles are promising drug carriers for targeted therapies, diagnostic imaging, and advanced vaccines. However, their clinical translation is limited by complex biological barriers that reduce cellular uptake and efficacy. In particular, the interaction with the cellular membrane controls nanoparticle adhesion, wrapping, or full engulfment, which ultimately governs nanoparticle internalization efficiency.
Flexible nanocarriers (e.g., liposomes, polymeric nanogels, and micelles) are particularly attractive because their deformability could help them enhance the probability of successful cellular entry. To understand the physical mechanisms associated with cellular uptake, we investigate the interaction of semi-flexible nanocarriers with a symmetric lipid bilayer using coarse-grained simulations.
We represent a flexible nanoparticle using the previously introduced metaparticle model and the membrane using the Cooke–Deserno model. By systematically varying nanoparticle properties, i.e., adhesion strength and topology, we identify distinct interaction regimes ranging from surface adhesion and trapping to complete wrapping and endocytosis. These regimes correlate with nanoparticle shape, size, and surface properties, providing quantitative design principles for optimizing cellular uptake.
Overall, this framework offers predictive insight into how the interplay between nanoparticle properties and membrane interaction governs cellular internalization, informing the rational design of next-generation soft nanocarriers and smart materials.
Paper details
Massimiliano Paesani, Ioana M. Ilie: Physical mechanisms of nanoparticle–membrane interactions: A coarse-grained study. J. Chem. Phys.164, 084906 (2026). DOI: 10.1063/5.0310512