Light reveals hidden radical chemistry of imidazolium salts
12 May 2026
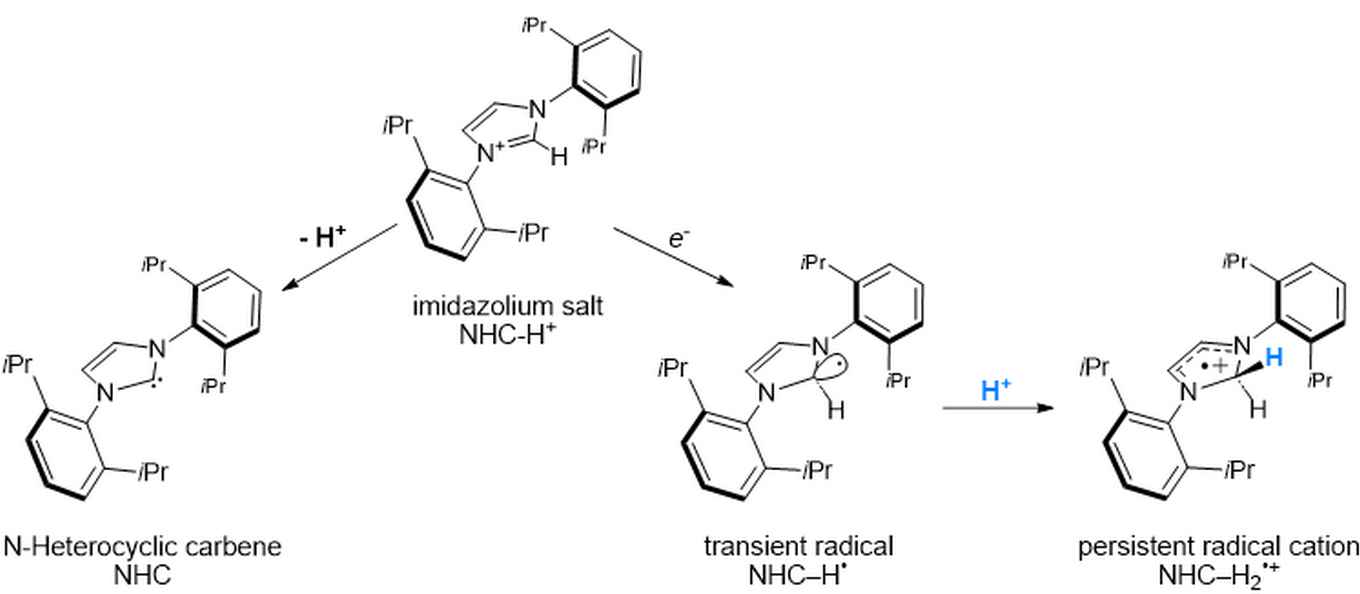
Imidazolium salts are widely used as precursors to N-heterocyclic carbenes (NHCs), which are important compounds in organometallic chemistry, catalysis, and organic synthesis. This conversion typically involves the removal of a proton. Whether imidazolium salts can also undergo single-electron processes under mild conditions has remained much less explored. The new study now shows that imidazolium salts are not merely passive precursors to NHCs, but can also act as redox-active platforms for radical chemistry.
From fleeting radicals to persistent species
The researchers used visible light to trigger electron transfer between imidazolium salts and electron-rich organic donors. This made it possible to observe short-lived NHC–H• radicals and follow their conversion into much more persistent NHC–H2•+ radical cations. The resulting species are unusual because their stability does not rely on an added external stabilizing group, but arises from the electronic structure of the imidazolium core itself.
A key insight from the study is that these persistent radical cations closely resemble the classical cyclohexadienyl radical. This analogy helps explain why NHC–H2•+ can survive long enough to be studied in detail: it’s stabilized by hyperconjugation within the imidazolium core.
The work provides new fundamental insight into the chemistry of imidazolium salts and expands the role of NHC chemistry into controlled open-shell reactivity. It also shows how light-driven electron transfer can be used to access reactive intermediates that are difficult to generate by conventional methods.
Abstract of the paper
Imidazolium salts are ubiquitous precursors to N-heterocyclic carbenes (NHCs), yet their radical chemistry remains largely unexplored, and structurally well-defined imidazolium-derived radicals are rare. Here, we show that visible-light excitation of charge-transfer (CT) bands of electron donor–acceptor (EDA) complexes between imidazolium cations and a triarylamine donor induces a single-electron transfer. Variable-temperature EPR spectroscopy reveals that irradiation at 100 K affords neutral [NHC─H]• radicals, which undergo protonation upon warming to room temperature to give the persistent σ-complex cations [IDippH2]•+ and [IXylH2]•+. Analysis of hyperfine couplings, in combination with density functional theory (DFT) calculations, establishes a close electronic analogy between [NHC─H2]•+ and the classical cyclohexadienyl radical [C6H7]•, highlighting σ–π hyperconjugation as the dominant stabilization motif. These findings identify imidazolium-derived σ-complex radicals as a unique class of carbene-based open-shell species and showcase imidazolium cations as noninnocent, redox-active platforms for controlled radical generation under mild photochemical conditions.
Paper details
Filipp M. Kolomeychuk, Lars J. C. van der Zee, Simon Mathew, Bas de Bruin, and J. Chris Slootweg: From [NHC-H]• to Persistent σ-Complex Radicals: Photoinduced Radical Chemistry of Imidazolium Salts. Angewandte Chemie International Edition, e5517752, 2026. DOI: 10.1002/anie.5517752