Charge-based separation of acid-functional polymers
21 April 2021
The research was performed in cooperation with the company Covestro (recently acquiring DSM Resins and Functional Materials business), one of the world’s key players in the field of water-borne coatings.
Abstract
Water-borne polymers are in ever-increasing demand due to their favourable ecological profile compared to traditional solvent-borne polymer systems. Many water-borne polymer particles are stabilized in aqueous media by the incorporation of acid-functional monomers. Due to the large variety of comonomers applied, these water-borne polymers have various superimposed statistical distributions, which make it challenging to obtain in-depth information regarding incorporation of the acidic monomers.
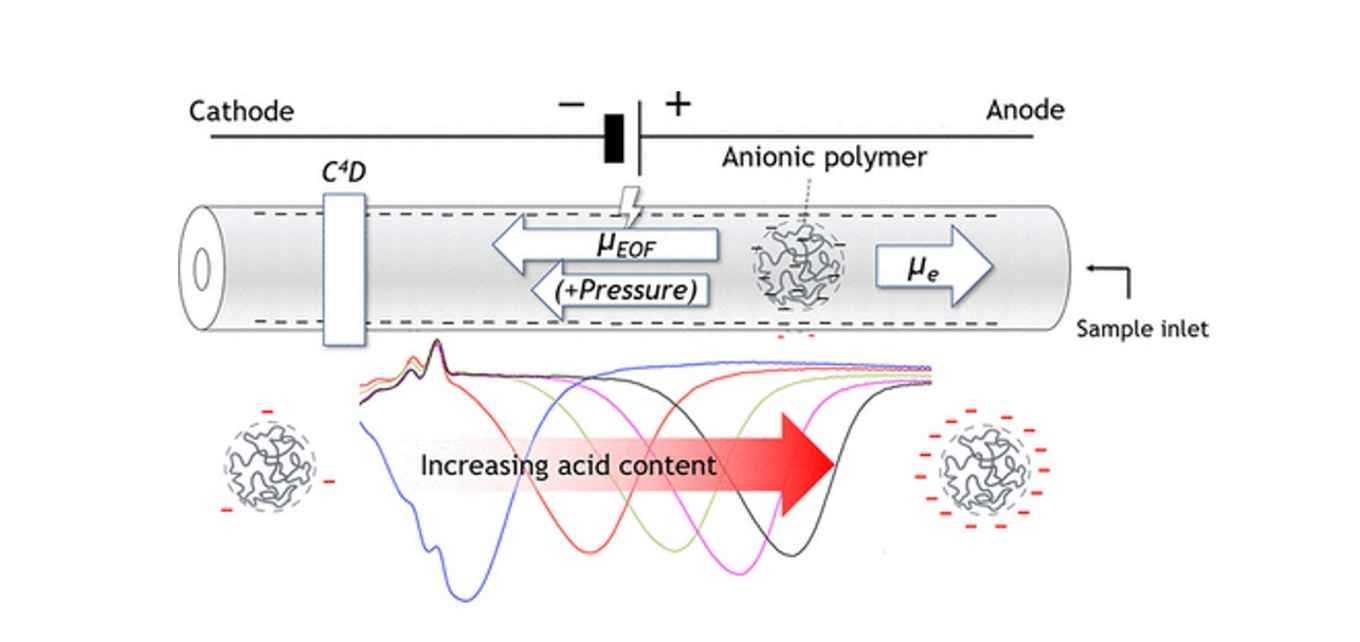
For selective analysis of the incorporated acidic monomers, a charge-based non-aqueous capillary electrophoresis (NACE) separation was developed. Two approaches were developed:
- deprotonation of the acid functionality with an organically soluble strong base; and
- heteroconjugation of anions of carboxylic acids with incorporated acid functionality.
In both approaches, N-methylpyrrolidone, as a strong solvent for polymers with a favourable relative permittivity for the presence of dissociated ionic species, was used for the separation.
It was shown that anions of carboxylic acids specifically associate with the incorporated acid groups in the polymers, resulting in negatively charged complexes that could be separated based on charge-to-size ratio by NACE. Although both approaches give comparable results with respect to acid distribution for acid-functional polymers, the effective mobility of the deprotonated polymers is roughly double that obtained from the heteroconjugation approach. Unlike the heteroconjugation approach, deprotonation conditions were detrimental to the fused-silica capillary, limiting practical use. Polymers with different chemical compositions, molecular weights, and acid contents were subjected to the CE approaches developed. Polymers with varying molecular weight but similar relative acid monomer content were shown to have similar migration times, which confirms that this approach separates polymers based on charge-to-size ratio.
Paper
Ton Brooijmans, Pascal Breuer, Aniek Schreuders, Myrthe van Tilburg, Peter J. Schoenmakers, and Ron A.H. Peters: Charge-Based Separation of Acid-Functional Polymers by Non-aqueous Capillary Electrophoresis Employing Deprotonation and Heteroconjugation Approaches. Analytical Chemistry 2021, 93, 14, 5924-5930. DOI: 10.1021/acs.analchem.1c00311
Links
- Research group Analytical Chemistry
- Website Centre for Analytical Sciences Amsterdam (CASA)