First synthetic galectin mimic as a platform for carbohydrate binding molecules
11 May 2021
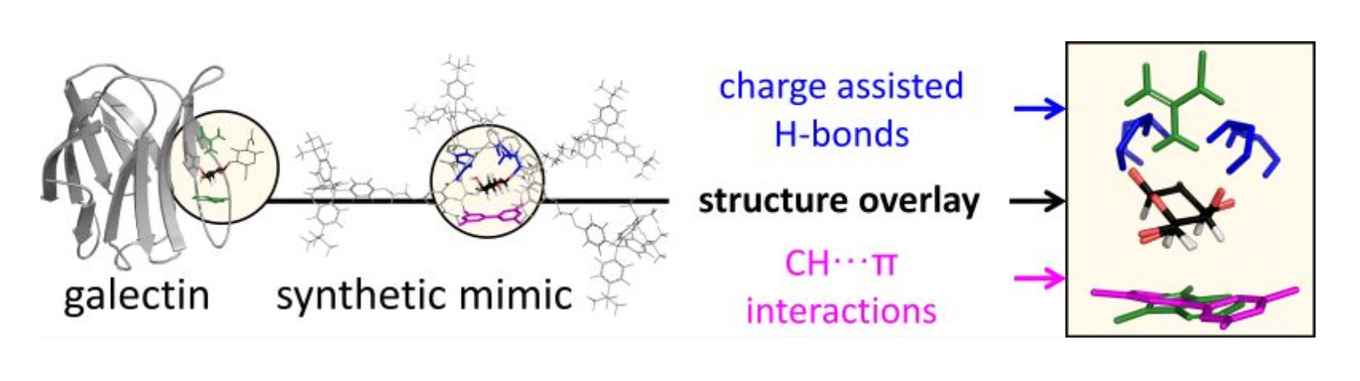
Developing the galectin mimic was a major part of Tiddo Mooibroek's Vidi research focused on the supramolecular chemistry of carbohydrates. Carbohydrates are widely regarded as an important class of biomolecules. Their selective detection and/or functionalization can be exploited for diagnostic and pharmacological purposes, and carbohydrates are a promising future renewable feedstock for the chemical industry.
Abstract
Galectins are a galactoside specific subclass of carbohydrate binding proteins (lectins) involved in various cellular activities, certain cancers, infections, inflammations and many other biological processes. The molecular basis for the selectivity of galectins is well‐documented and revolves around appropriate interaction complementarity: an aromatic residue for C–H···π interactions and polar residues for (charge assisted) hydrogen bonds with the axial hydroxyl group of a galactoside. Despite this knowledge, no synthetic mimics are currently available. Here, we report on the design and synthesis of the first galectin mimic (6), and show that it has a higher than 65‐fold preference for n‐octyl‐β‐galactoside (8) over n‐octyl‐β‐glucoside (7) in CD2Cl2 containing 5% DMSO‐d6 (with Ka ≥ 4500 M‐1 for 6:8). Molecular modelling informed by nOe studies reveal a high degree of interaction complementarity between 6 and galactoside 8, which is very similar to the interaction complementarity found in natural galectins.
Paper
Tiddo Jonathan Mooibroek, Brian Timmer, Xander Schaapkens and Arjaan Kooijma: A synthetic galectin mimic. Angew. Chem. Int. Ed. First published: 08 May 2021. DOI: 10.1002/anie.202104924