Highly efficient meta-C−H arylation of anisole derivatives
6 September 2022
Abstract
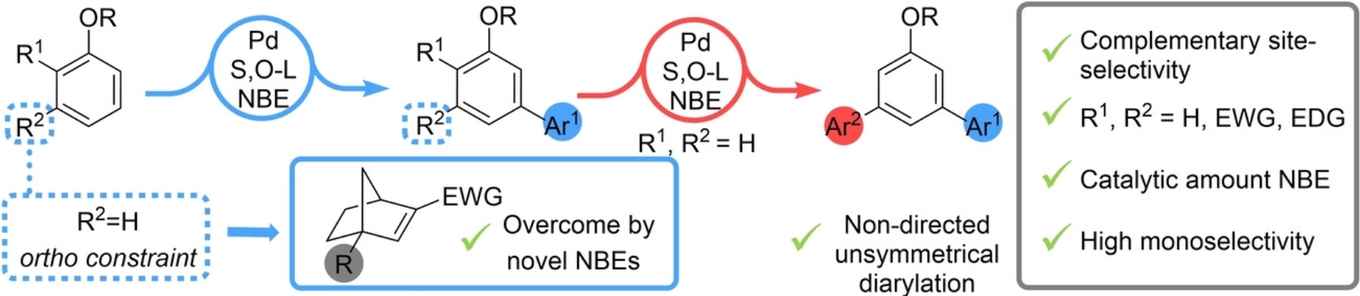
Reversing the conventional site-selectivity of C−H activation processes provides new retrosynthetic disconnections to otherwise unreactive bonds. Here, we report a new catalytic system based on palladium/norbornene and an S,O-ligand for the meta-C−H arylation of aryl ethers that significantly outperforms previously reported systems. We demonstrate the unique ability of this system to employ alkoxyarene substrates bearing electron donating and withdrawing substituents. Additionally, ortho-substituted aryl ethers are well tolerated, overcoming the “ortho constraint”, which is the necessity to have a meta-substituent on the alkoxyarene to achieve high reaction efficiency, by enlisting novel norbornene mediators. Remarkably, for the first time the monoarylation of alkoxyarenes is achieved efficiently enabling the subsequent introduction of a second, different aryl coupling partner to rapidly furnish unsymmetrical terphenyls. Further insight into the reaction mechanism was achieved by isolation and characterization of some Pd-complexes—before and after meta C−H activation—prior to evaluation of their respective catalytic activities.
In a short profile in Angewandte Chemie, research leader Tati Fernández-Ibáñez comments on this research: “We were working for a while on the combination of our Pd/ S,O-ligand catalyst and norbornene with the aim to enable new retrosynthetic disconnections for arenes. We found that by modulating the structure of the S,O-ligand as well as the norbornene we have developed a really efficient methodology for the meta-arylation of anisole derivatives. We are still surprised with the experiment that proved that the C–H activation is not the rate-limiting step, as usually it is”.
Paper details
Verena Sukowski, Manuela van Borselen, Dr. Simon Mathew, Prof. Dr. M. Ángeles Fernández-Ibáñez: S,O-Ligand Promoted meta-C−H Arylation of Anisole Derivatives via Palladium/Norbornene Catalysis. Angew. Chem. Int. Ed. 2022, 61, e202201750. DOI: 10.1002/anie.202201750
See also
- Introducing M. Ángeles (Tati) Fernández-Ibáñez (Angewandte Chemie author profile)
- Website Fernández-Ibáñez research group