How a formyl group makes molecular motors more efficient
30 April 2024
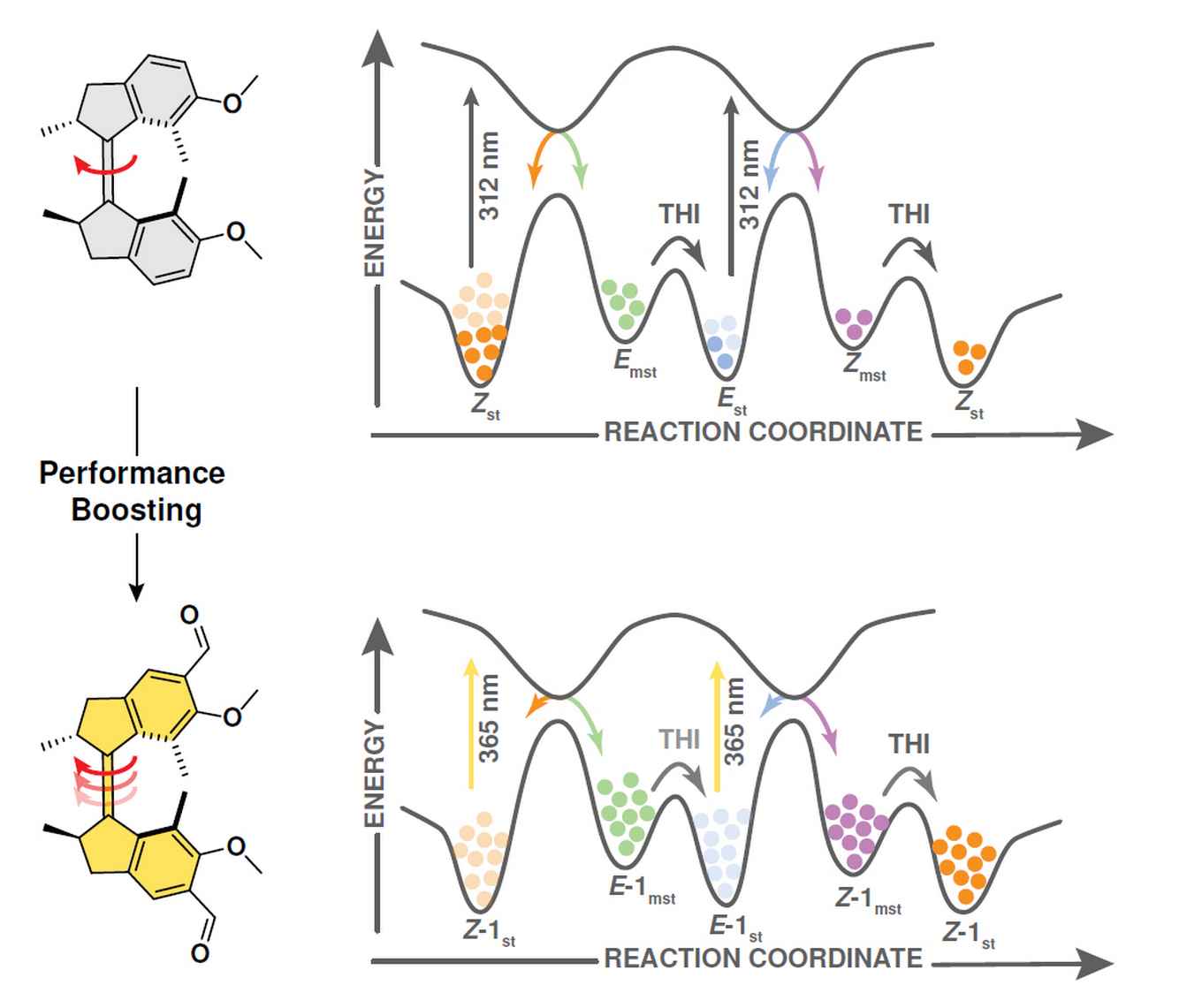
The research was aimed at so-called overcrowded alkene-derived molecular motors which constitute the first generation of molecular motors developed by the Feringa group. These convert light and heat into chirality-directed unidirectional rotary motion, albeit with a rather low efficiency. The Nature Chemistry paper shows how appending a formyl group in the position directly conjugated with the central alkene bond (the axle of rotation) induces a red shift of the absorption and, more importantly, boosts photoisomerization quantum yields above 60%. This surpasses the efficiency of artificial light-driven motors commonly applied in the past decades and is comparable to the most efficient retinal-opsin visual pigment - a benchmark of efficiency for photoinduced double-bond isomerizations.
To provide more insight into the remarkable photoisomerization behaviour, Paradiz Domínguez, Sardjan and Buma performed femtosecond and nanosecond transient absorption (TA) measurements and contributed to time-dependent DFT calculations to map the electronic decay pathways. They show, among others, the existence of a long-lived (decaying on a tens of nanosecond time scale) exited state that is tentatively associated with the lowest triplet state. Furthermore, they identify two optically dark states with nπ* character localized on the aldehyde moieties at lower energies. Based on these observations, they propose an intricate working mechanism for the molecular motor. They also demonstrate that the altered photochemistry cannot only be attributed to the added formyl group, as initially seemed to be suggested by the observed contribution of a triplet-mediated decay pathway. Instead, it is the cooperative effect of the carbonyl moiety with details of stator and rotor that determine the contribution of the triplet-mediated decay pathway.
Abstract of the paper
Artificial molecular motors and machines constitute a critical element in the transition from individual molecular motion to the creation of collective dynamic molecular systems and responsive materials. The design of artificial light-driven molecular motors operating with high efficiency and selectivity constitutes an ongoing fundamental challenge. Here we present a highly versatile synthetic approach based on Rieche formylation, which boosts the quantum yield of the forward photoisomerization reaction while reaching near-perfect selectivity in the steps involved in the unidirectional rotary cycle and drastically reducing competing photoreactions. This motor is readily accessible in its enantiopure form and operates with nearly quantitative photoconversions. It can easily be functionalized further and outperforms its direct predecessor as a reconfigurable chiral dopant in cholesteric liquid crystal materials.
Paper details
Jinyu Sheng, Wojciech Danowski, Andy S. Sardjan, Jiaxin Hou, Stefano Crespi, Alexander Ryabchun, Maximilian Paradiz Domínguez, Wybren Jan Buma, Wesley R. Browne, Ben L.Feringa: Formylation boosts the performance of light-driven overcrowded alkene-derived rotary molecular motors. Nature Chemistry (2024). DOI: 10.1038/s41557-024-01521-0
See also
- University of Groningen news item: More efficient molecular motor widens potential applications
- Molecular Photonics research group