Optimised borophane nanosheets for hydrogen storage
Doping with potassium significantly enhances performance
23 March 2026
Borophane is essentially a hydrogenated form of 2D borophene, a graphene-like material consisting of boron instead of carbon. Borophane is expected to store and release hydrogen at relatively mild, near-ambient conditions. However, experimental studies in this direction are limited.
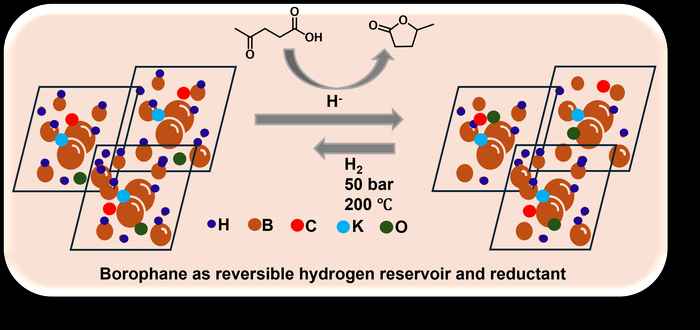
The study reported in Small addresses this gap by synthesizing potassium-doped borophane (BH) nanosheets and evaluating their performance as hydrogen storage materials. Potassium plays a crucial role in modulating the electronic structure of the BH lattice, enhancing hydrogen binding and activation through charge polarization effects. The doped BH sheets exhibit reversible hydrogen storage behavior, with partial regeneration achieved under hydrogen pressures of 50 bar. Density functional theory (DFT) calculations support these findings, predicting a theoretical hydrogen storage capacity of approximately 4.2 wt%. Experimental analyses further reveal that hydrogen is released at relatively mild temperatures, and only specific B-H sites participate in the storage and release process, highlighting a site-selective mechanism.
In addition to hydrogen storage, the study demonstrates the dual functionality of potassium-doped borophane as a metal-free reducing agent through selective hydrogen transfer. The BH nanosheets effectively facilitate the conversion of levulinic acid (LA), a biomass-derived platform molecule, into γ-valerolactone (GVL), which typically requires high temperatures and high hydrogen pressures or noble metal catalysts. Using borophane as a hydrogen provider, the conversion could be carried out at significantly milder conditions.
The study thus provides experimental validation for alkali-metal-modified borophanes acting as a multifunctional material for hydrogen storage and transfer, opening avenues for sustainable energy and other applications.
The research was part of the project ‘2D BoroCat’ led by Dr. Anju Rajamohanan Sobhana as a Marie Curie postdoctoral fellow at the HIMS Catalysis Engineering group. It was carried out in collaboration with researchers at the Indian Institute of Technology in Roorkee and Mumbai, TU Delft, Tata Steel, and Vrije Universiteit Amsterdam (VU). The paper is dedicated to the memory of VU co-author Prof. Freek Ariese, who suddenly passed away in early January 2026.
Paper details
Rajamohanan Sobhana Anju, Pankaj Kumar, Dhanaji R. Naikwadi, Bettina Baumgartner, Savi Chaudhary, Atul Bansode, Merel C. Konings, Freek Ariese, Erdni D. Batyrev, Prasad Gonugunta, Vimal Chandra Srivastava, Ramaswamy Murugavel, N. Raveendran Shiju: Potassium-Doped Borophane Nanosheets: A Multifunctional Platform for Reversible Hydrogen Storage and Metal-Free Hydrogen Transfer Small, 2026, Early View e11090. DOI: 10.1002/smll.202511090